Company Profile
Metagone is a clinical stage biotech company, focuses on innovative drug research and development which is novel targets and small molecule drugs.
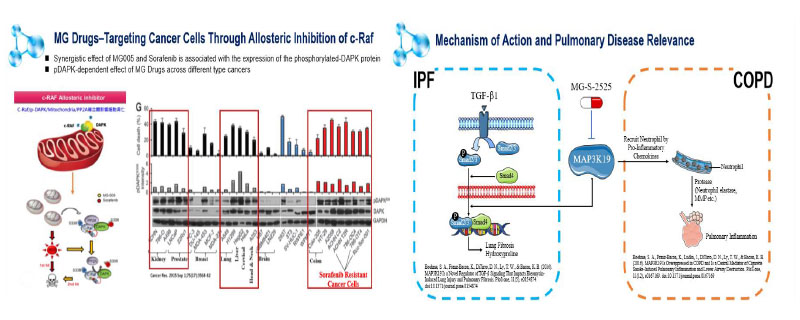
The MG agnostic-tumor anticancer is oral, new combination treatment which including the first generation MG-D-1509 and the second generation MG-D-1609. The mechanism of action indicates allosterically target to c-Raf and specifically associate with the pDAPK of tumor cells leaving mitochondria produce high amounts of ROS and subsequent pDAPK dephosphorylation reaction. The MG-D-1509 was completed Phase I clinical trials. Followed by the new salts of MG-D-1609 increases the solubility and reduces the dosage. With the support of MG-D-1509 phase I result, the MG-D-1609 is completed phase I trial and slating into phase II trial with 4 types of cancer (liver, lung, kidney, colorectum). The pDAPK biomarker will help our Phase II tumor-agnostic basket trial in solid tumors.
The MG-S-2525 new drug is an oral, small molecule, anti-inflammatory agent. Its active ingredient HL0 is a newly discovered mitogen-activated protein kinase 19 (MAP3K19) inhibitor. The novel and high-performance RNA of MAP3K19 was discovered from macrophages in the alveolar lavage fluid from IPF patients, COPD patients and healthy smokers and confirmed this high specificity of MAP3K19 protein only on lung tissue. In the bleomycin-induced pulmonary fibrosis animal model, it was observed that the MG-S-2525 new drug can effectively control fibrosis and was superior than Roche’s Esbriet. MG-S-2525 new drug obtained the Orphan Drug Designation (ODD) for IPF disease from the US FDA. The phase I results showed the MG-S-2525 drug in the maximum dose of 800 mg was highly safe, and the pharmacokinetic parameters was correlated with ascending doses. The MG-S-2525 is open for IPF phase IIa trial with studying efficacy-related biomarker. Also, in the vitro study, the MG-S-2525 has the better reduction of inflammation up to 50% than Azithromycin in the Bronchiectasis.
Product / Service
- Anticancer NCE treats on agnostic tumor with pDAPK biomarker expression, is conducting phase II clinical trial
- NCE treats IPF with MAP3K19 novel target, is conducting phase IIa trial
- NCE treats COPD with MAP3K19 novel target is open for phase IIa trial
- First in class drug
- Focus on Oncology and Respiratory drug development field
Business Interests
R&D Collaboration, Licensing
Contact Info
Maggie Lu / Simon Huang
General Manager / Executive Vice President
maggie@metagone.com.tw / simon@metagone.com.tw
886-2-2790-6566 ext. 201 / 886-2-2790-6566 ext. 203